Pharmaceutical
Functional lipid solubilizers, emulsifiers and surfactants to enhance the bioavailability of insoluble and poorly permeable Active Pharmaceutical Ingredients (APIs). All our lipid excipients are highly-reproducible, monograph compliant and manufactured in ISO certified facilities in accordance with cGMP and IPEC guidelines.

Self-emulsifying Drug Delivery
Emulsifiers, solubilizers and surfactants to enhance oral delivery and absorption of lipophilic drugs.
Transdermal Drug Delivery
Functional lipids for emulsified topical delivery systems, incorporating insoluble actives into creams or gels.

Solubilization
Enhance solubility for increased bioavailability of BCS Class II and Class IV actives.
Permeation Enhancement
Enhance permeability in oral, topical, transdermal and parenteral drug delivery systems.

Sustained Release
Functional lipids and hard fats for matrix and encapsulated dosage forms in sustained release applications.
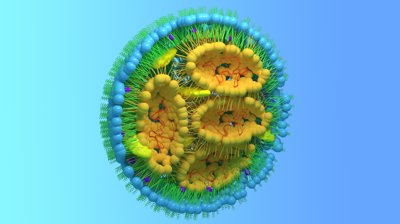
LNP Therapeutics
c-GMP certified custom lipids for nucleic acid delivery and improved efficacy in biopharma applications.
Direct Compression
Functional lipids and co-processed excipient systems for direct compression of liquid lipids and other applications.

Lubrication
Lipid excipients for the lubrication of soft gels and direct compression tablets.
Product Search
If you already know what product you need, just search here.

