CAPMUL® GDB EP/NF
Formulating high doses of sustained release BCS I drugs can be extremely challenging especially at the risk of compromising stability, or other vital parameters needed to develop a successful solution. ABITEC's CAPMUL® GDB EP/NF (glyceryl dibehenate) is a versatile lipid for sustained release applications where these barriers can be overcome and at the advantage of improved overall performance. In addition, it is prominently utilized in a range of other processes across the tableting space such as, direct compression, rapid mix granulation, and tablet lubrication.

Watch our short video introduction to CAPMUL® GDB EP/NF applications
CAPMUL® GDB EP/NF Application Uses
Properties
CAPMUL® GDB EP/NF Properties
Description
CAPMUL GDB EP/NF is a mixture of diglycerides, mainly glyceryl dibehenate, together with variable quantities of monoglycerides and triglycerides. It is obtained by the esterification of glycerol with behenic acid of vegetable origin.
Composition
CAPMUL GDB EP/NF consists predominately of diglycerides (40 – 60 %) made from behenic acid. Behenic acid has 21 carbons plus a carboxyl group, and is fully saturated, i.e. no double bonds (C22:0). The remainder of the mixture is composed of monoglycerides (15 – 23%) and triglycerides (21 – 35%).
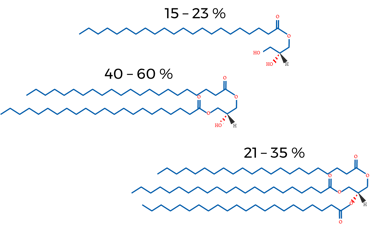
Physiochemical Properties
CAPMUL GDB EP/NF is manufactured by spray congealing molten lipid which produces a particle size of ≤ 125 µm as determined by a sieve analysis. The melting point of CAPMUL GDB EP/NF is between 63 – 77 °C as determined by Differential Scanning Calorimetry (DSC). CAPMUL GDB EP/NF recrystallizes almost immediately upon reaching a freezing point of 70 °C.
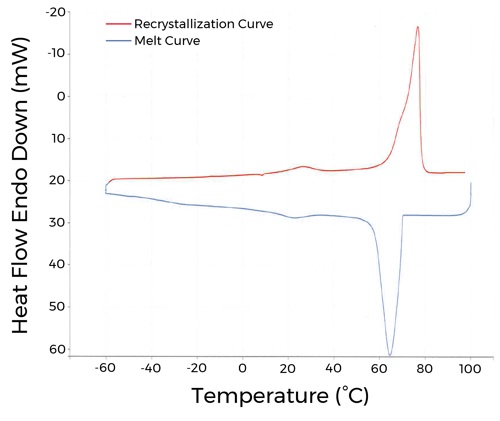
Figure 1. CAPMUL GDB EP/NF melting and crystallization thermograms as determine by DSC at heating and cooling rates of 10.00 °C/min.
Regulatory Status
CAPMUL GDB EP/NF is manufactured to comply with both the monographs for the European Pharmacopeia (EP) as well as the United States Pharmacopeia and National Formulary (USP-NF) as shown in the table below.
Pharmacopeia |
Description |
|
USP – NF |
Glyceryl Dibehenate |
|
EP |
Glycerol Dibehenate |
Unit Operations
CAPMUL® GDB EP/NF in Pharmaceutical Unit Operations
CAPMUL GDB EP/NF is a highly-functional, highly-reproducible solid lipid excipient manufactured for use in pharmaceutical formulations. CAPMUL GDB EP/NF meets the specifications of both the EP and NF for glyceryl dibehenate.
CAPMUL GDB EP/NF is a versatile excipient that is readily employed in a multitude of pharmaceutical manufacturing unit operations to providing varying functionalities, including multi-particulate diluent, sustained-release coating, matrix sustained-release excipient, tablet lubricant, protective coating, and taste-masking excipient.
CAPMUL GDB EP/NF is readily employed in the following pharmaceutical unit operations:
- direct compression tableting
- granulation/hot-melt extrusion
- spray congealing
- spray coating
- solid lipid nanoparticle manufacturing
Direct compression tableting
CAPMUL GDB EP/NF is generally employed in direct-compression tableting as a sustained- release matrix excipient or as a boundary lubricant. In both cases, the CAPMUL GDB EP/NF is added directly into the tablet formulation by blending with other formulation ingredients, employing typical industry mixing equipment. The physiochemical properties, shape, and particle size of CAPMUL GDB EP/NF allow the excipient to be readily dispersed in a uniform fashion, in order to produce a randomized mix with the remaining tableting ingredients upon blending.
Figure 1: Manufacturing unit operations for direct compression with CAPMUL GDB EP/NF
With regard to sustained release, the concentration of CAPMUL GDB EP/NF within each respective formulation will control the release of the active. Generally, as more CAPMUL GDB EP/NF is added to the formulation, the slower the dissolution rate. The primary mechanism of active dissolution out of a sustained release tablet manufactured with CAPMUL GDB EP/NF is diffusion. As such, selection of diluent excipients will affect the active dissolution rate, with water soluble diluents, such as lactose and mannitol increasing dissolution rate, as compared with water insoluble excipients such as dibasic calcium phosphate, which reduce dissolution rate, comparatively.
CAPMUL GDB EP/NF is a boundary lubricant, indicating it does not shear and spread over tablet ingredient surfaces upon blending. In contrast, the CAPMUL GDB EP/NF diffuses uniformly throughout the tablet blend upon mixing. In this way, at points of contact between the CAPMUL GDB EP/NF and tablet machinery, such as the die walls and punches, the CAPMUL GDB EP/NF provides boundary lubrication, reducing sticking and lowering ejection force.
Granulation
CAPMUL GDB EP/NF can be employed in aqueous, solvent-based, and hot-melt granulations. In both aqueous and solvent-based granulations, CAPMUL GDB EP/NF is typically employed as a sustained-release excipient. CAPMUL GDB EP/NF can be added as part of the granulation unit operation, or it can be added after granulation prior to direct compression as lubricant for direct compression of the granules. Active release can be adjusted by adjusting the concentration of CAPMUL GDB EP/NF in the granule formulation.
Figure 2: Manufacturing unit operations for granulations with CAPMUL GDB EP/NF
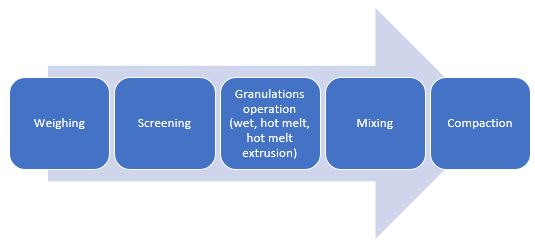
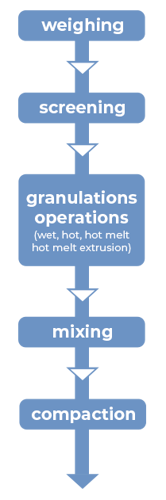
In hot-melt granulations, including hot-melt granulations manufactured by hot-melt extrusion, CAPMUL GDB EP/NF can act as a binder, a taste-masked coating, a protective coating, a diluent, and/or a sustained-release excipient. For hot melt extrusion, the CAPMUL GDB EP/NF is blended with other formulation excipients in concentrations suitable for application purposes and this mixture is introduced into a hot-melt extruder. Subsequently, the granulation or extrudate is collected for further processing. CAPMUL GDB EP/NF can be employed in other hot-melt unit operations, such as high shear hot-melt granulation which employs a jacketed hi-shear granulator or in hot melt fluid bed granulations where CAPMUL GDB EP/NF is sprayed onto a fluidized bed of active and other granulation excipients. Generally, the extrudates or granules from these operations are sized and then blended with other tableting excipients and then compacted on rotary tableting equipment.
Spray congealing/drying
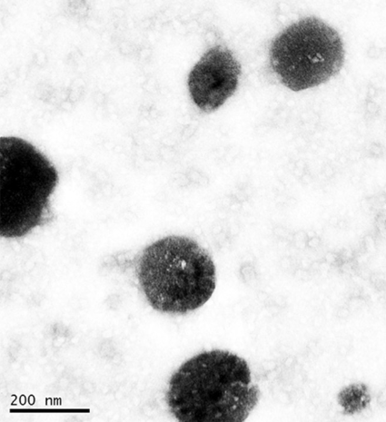
CAPMUL GDB EP/NF is a solid lipid excipient at room temperature. As such, it is readily employable in spray congealing unit operations. CAPMUL GDB EP/NF can be molten with lipid soluble excipients to form a molten mixture, which can be sprayed into lipid/active multi-particulates for subsequent processing (Figure 3). Additionally, lipid insoluble actives can be uniformly suspended in CAPMUL GDB EP/NF. This lipid/active suspension can be sprayed to form multi-particulates for subsequent processing. In a spray congealing unit operation, CAPMUL GDB EP/NF can be employed primarily as a sustained-release excipient, a taste-masking excipient, a multi-particulate diluent, or a protective coating against chemical reactions, such as oxidation or hydrolysis.

Figure 3: Ibuprofen and glyceryl behenate solid lipid nanoparticle (1)
Spray Coating
CAPMUL GDB EP/NF can be employed as a functional coating for hot-melt, fluid-bed unit operations. CAPMUL GDB EP/NF alone or with other ingredients can be sprayed in molten form onto a fluidized bed of raw materials, including actives and other excipients. This unit operation generates coated multi-particulates for subsequent processing. In this application, CAPMUL GDB EP/NF can be employed as a sustained release coating, a protective coating, or taste-masking coating.
Solid lipid nanoparitcle manufacturing (2)
CAPMUL GDB EP/NF mixed with select surfactants can form spontaneous self-emulsifying active-carrying isotropic systems when homogenized or stirred in hot water. Upon cooling and solvent evaporation, these micellar systems form active carrying solid lipid nanoparticles (Figure 4), which can be employed in the further processing of a dosage forms. This type of processing allows for specialized routes of administration, such as inhalation. Additionally, active contained in the core of the lipid nanoparticle is protected from oxidation and hydrolysis events.
Figure 4: Rifabutin containing solid lipid nanoparticles manufactured with glyceryl dibehenate
References
- Priscilla Chui Hong Wong, Paul Wan Sia Heng & Lai Wah Chan (2016) A study on the solid state characteristics of spray-congealed glyceryl dibehenate solid lipid microparticles containing ibuprofen, Drug Development and Industrial Pharmacy, 42:3, 364-377
- Gaspar D. et al (2016) Rifabutin-loaded solid lipid nanoparticles for inhaled antitubercular therapy: Physicochemical and in vitro studies, International Journal of Pharmaceutics, 497, 199-209.
Release Mechanism
CAPMUL® GDB EP/NF Drug Release Mechanism
Obtaining the desired sustained-release profile for a therapeutic is often a challenge many formulators face. There are many options for manufacturing sustained-release tablets, but one of the most common approaches is matrix sustained release. With regard to the manufacturing of matrix sustained-release tablets, two common types of matrices are employed, (1) swelling/eroding matrices and (2) non-swelling/non-eroding matrices. The swelling/eroding matrix approach incorporates the active in a swellable, water-soluble polymer, with drug-release controlled by diffusion of the active through the swelling polymer and also by drug-release occurring from the erosion of the polymer. The non-swelling/non-eroding matrix approach incorporates the active in a water-insoluble matrix, which does not swell and does not erode, or erodes very slowly, with drug release controlled primarily by diffusion of the active out of the insoluble matrix (Figure 1).
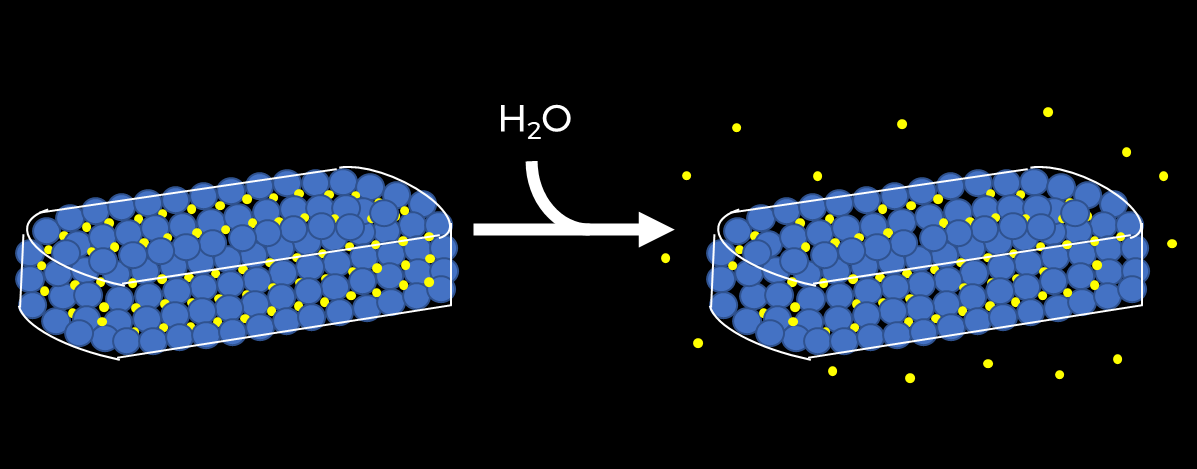
Figure 1. Representation of a diffusion-controlled drug release
Commonly used drug release water-soluble excipients include polymers like Hypromellose (i.e. HPMC). Methylcellulose polymers like Hypromellose function by absorbing water, swelling, and slowly releasing the therapeutic by either diffusion or erosion of the swelled gel matrix. A number of polymer parameters must be taken into account when formulating with a product like HPMC. The polymer viscosity, polymer molecular weight and drug/polymer ratio, as well as numerous other factors can all affect how the therapeutic is released from the tablet, making the process of formulating a desired release-profile that much more difficult.


Figure 2. Physical appearance of a sustained release tablet made with CAPMUL GDB EP/NF. Data is courtesy of IMCD Pharma Technical Centre PTC, Mumbai.
The release mechanism for CAPMUL GDB EP/NF Is based solely on diffusion as CAPMUL GDB EP/NF is a water-insoluble excipient. Unlike HPMC, CAPMUL GDB EP/NF does not erode. This allows for the formulation of a tablet that remains undisturbed after dissolution (Figure 2). Because CAPMUL GDB EP/NF is less sensitive to pH and is insoluble to water, the primary parameter which needs to be optimized in the formulation is the CAPMUL GDB EP/NF concentration, allowing for a simple product development process.
Optimizing Drug Release with Capmul GDB EP/NF
Obtaining a desired drug release profile is dependent on many factors including the manufacturing process (i.e. HME, direct compression, spray drying, etc…), diluents, and most importantly, the concentration of the sustained release excipient employed in the formulation. When utilizing Capmul GDB EP/NF as the sustained-release excipient, Formulators can adjust the drug-release profile by simply modifying the concentration of Capmul GDB EP/NF in the final drug product (FDP). The more Capmul GDB EP/NF added to the FDP, the slower the drug release (Figure 3). As indicated above the exact sustained-release profile will be dependent on drug solubility, dose, tablet surface area, dissolution media, and other excipients in the formulation, such as diluents, binders, and lubricants.
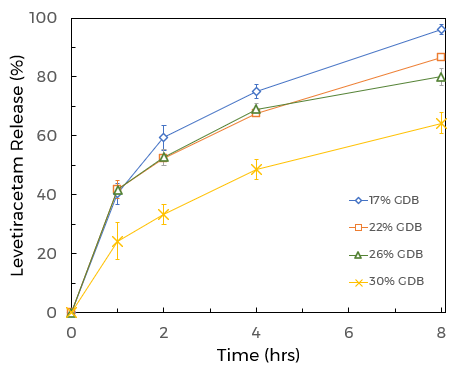
Figure 3. CAPMUL GDB concentration-dependent release of a high does BCS class I molecule. Data is courtesy of IMCD Pharma Technical Centre PTC, Mumbai.
Tablet Characteristics
CAPMUL® GDB EP/NF Tableting Characteristics
CAPMUL GDB EP/NF is a highly-functional, highly-reproducible solid lipid excipient manufactured for use in pharmaceutical formulations. CAPMUL GDB EP/NF meets the specifications of both the EP and NF for glyceryl dibehenate.
The most commonly measured physical tableting characteristics are weight, thickness, hardness, and friability. Uniformity and reproducibility on these physical characteristics are important for the production of a quality and uniform tablet dosage form. Consistent thickness is often important to subsequent packaging operations, while hardness and friability are indicators of tablet durability. Once of the most important characteristics of any tableting excipient is compactability, which is the measure of hardness at specific compression pressures (Table 1).
Table 1: Comparison of tableting characteristics of CAPMUL GDB EP/NF with a competitive glyceryl dibehenate in matrix tablet formulation of levetiracetam, dibasic calcium phosphate, and magnesium stearate. Data is courtesy of IMCD Pharma Technical Centre PTC, Mumbai.
| Compressed Tablet Parameter | CAPMUL GDB (22%) Lipid Matrix Tablet | Competitor GDB (22%) Lipid Matrix Tablet |
| Weight (in mg) | 1110 | 1110 |
| Hardness (in N) | 115-130 | 72-83 |
| Thickness (in mm) | 6.65-6.78 | 6.66-6.75 |
| Friability (in %) | 0.15 | 0.12 |
| Punch Size (in mm) | 20 X 10 | 20 X 10 |
| Compression Force (kN) | 24-35 | 36-42 |
For a comparative study, CAPMUL GDB EP/NF and a competitive glyceryl dibehenate were used to create a sustained release tablet. As indicated in Table 1, thickness and friability between the two formulations were similar. However, the tablet formulation containing CAPMUL GDB EP/NF was significantly harder, indicating that CAPMUL GDB EP/NF is more compactable than the competitor glyceryl dibehenate. This allows tablet formulations employing CAPMUL GDB EP/NF to achieve greater hardness and durability at lower compression forces, which can improve press and tooling life. Additionally, formulations containing CAPMUL GDB EP/NF will form suitable tablets at lower compression forces and be less susceptible to over compression.
Lubrication
CAPMUL® GDB EP/NF for Lubrication
CAPMUL GDB EP/NF is a highly-functional, highly-reproducible solid lipid excipient manufactured for use in pharmaceutical formulations. CAPMUL GDB EP/NF meets the specifications of both the EP and NF for glyceryl dibehenate.
Lubrication on the rotary tablet press is highly important. If a tablet formulation is not optimized for lubrication, then sticking to punch face and die walls can occur, resulting in tablet defects, such as sticking and picking. Additionally, if a tablet blend is under lubricated, then upon ejection of the tablet from the die, high ejection forces can occur, resulting in the breaking of bonds formed during compaction, leading to tablet defects such as capping and lamination. In other cases, the use of traditional lubricants like magnesium stearate may affect the stability of certain actives and thus alternatives should be explored.
CAPMUL GDB EP/NF, due to is lipidic nature, can be employed as a boundary lubricant in pharmaceutical mixing unit operations. Unlike many traditional lubricants, CAPMUL GDB EP/NF does not smear over the surfaces of other tableting excipients, when exposed to the shearing forces that occur during blending. In contrast, CAPMUL GDB EP/NF, due to its physiochemical, shape, and size properties, readily diffuse uniformly throughout the mix during blending. Where CAPMUL GDB EP/NF touches tablet machinery surfaces, such as the die walls and punch faces, it provides lubrication.
The advantages of a boundary lubricant such as CAPMUL GDB EP/NF include increased tablet robustness, as contact points between diluents and other tablet binders are not occluded by the lubricant covering particle surfaces. Additionally, since CAPMUL GDB EP/NF does not smear over particle surfaces in the formulation, disintegration times are not as affected as with other lubricants, as water can readily contact and dissolve the non-lubricant tableting excipients. Furthermore, CAPMUL GDB EP/NF is not blend time sensitive, as it does not shear during mixing. In fact, CAPMUL GDB EP/NF does not require a separate lubricant blending unit operation and can be included in the primary blend with other tableting excipients.
Legal Disclaimer
CAPMUL® is a trademark of ABITEC Corporation. All information and statements given on this website are believed to be accurate at the time of publication. However, neither ABITEC Corporation nor any of their affiliates make any representations or warranty with respect thereto, including, but not limited to, any results obtained in the processing of the products by customers or any third party. All information and statements are intended for persons having the required skill and know-how and do not relieve the customer or user from verifying the suitability of information and statements given for a specific purpose prior to use of the products. It is entirely the obligation of the customer or user to comply with applicable laws and regulations, and also with all patent or other intellectual property rights of third parties. ABITEC CORPORATION EXPRESSELY DISCLAIMS ANY REPRESENTATIONS OR WARRANTIES OF ANY KIND, WHETHER EXPRESSED OR IMPLIED, AS TO THE ACCURACY, CURRENCY, COMPLETENESS AND/OR THE MERCHANTABILITY OR FITNESS OF A PARTICULAR PURPOSE OF ANY INFORMATION CONTAINED ON THIS WEBSITE AND/OR PRODUCT DESCRIBED OR PROMOTED ON THIS WEBSITE, INCLUDING WARRANTIES WITH RESPECT TO INFRINGEMENT OF ANY PATENT, COPYRIGHT, OR OTHER RIGHTS OF A THIRD PARTY. We reserve the right to change product specifications and specified properties of the products without prior notice.

